Bryan Johnson, a self-proclaimed “biohacker” and tech millionaire, is on a highly publicized and controversial quest to reverse the aging process. His extreme regimen, which he refers to as his “longevity protocol,” involves a multitude of unconventional practices, ranging from blood plasma transfusions from his teenage son to facial injections of donor fat. A central component of his protocol is a strict diet and exercise regime, coupled with the latest experimental treatments, including microdosing weight loss drugs typically prescribed for obesity and diabetes.
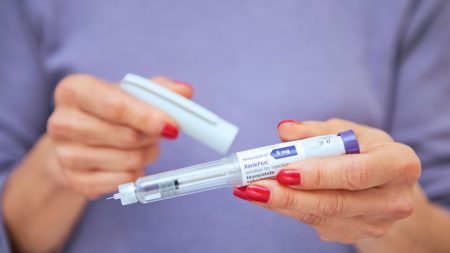
Johnson’s recent decision to incorporate microdosing of tirzepatide, a potent weight-loss drug, has sparked debate. Despite having minimal body fat and already being metabolically healthy, Johnson claims he’s using a reduced dose of the drug, marketed as Mounjaro, not for weight loss, but for its purported anti-aging properties. He cites emerging research suggesting that GLP-1 agonists, the class of drugs to which tirzepatide belongs, might have benefits beyond weight loss, such as anti-inflammatory effects, cardiovascular improvement, and cognitive preservation. He believes microdosing allows him to harness these potential benefits while minimizing the risk of side effects commonly associated with higher doses.
However, medical experts have raised concerns about Johnson’s self-experimentation and the potential risks associated with microdosing weight-loss drugs without medical supervision. Tirzepatide, like other GLP-1 agonists, works by mimicking a gut hormone that signals satiety to the brain, regulating blood sugar, and reducing appetite. While studies have explored the drug’s potential benefits for various conditions, including cardiovascular health and Alzheimer’s prevention, these studies are preliminary, and the long-term effects of microdosing in individuals without obesity or diabetes remain largely unknown.
Experts warn that even at lower doses, GLP-1 agonists can cause side effects, including nausea, vomiting, digestive issues, and, in rare cases, more serious complications like pancreatitis and gallbladder problems. They emphasize the importance of consulting with a healthcare professional before considering any weight-loss medication, particularly for individuals who are not obese or do not have diabetes. The potential for misuse and the unknown long-term effects underscore the need for caution and further research.
The availability of weight-loss drugs like Wegovy (semaglutide) and Mounjaro (tirzepatide) has generated significant interest, especially given anecdotal reports of dramatic weight loss. While these medications offer a promising new avenue for treating obesity, their efficacy and safety profiles need to be carefully evaluated, especially in the context of off-label use, like Johnson’s microdosing regimen. Experts advocate for a holistic approach to weight management, which includes lifestyle modifications, dietary changes, and psychological support, alongside medication, for optimal and sustainable results.
The emergence of these potent weight-loss drugs has also ignited a broader conversation about societal attitudes towards weight and the complex interplay of genetics, lifestyle, and environment in influencing body weight. While some view these drugs as a potential “game-changer” in addressing the obesity epidemic, others caution against over-reliance on medication without addressing the underlying factors contributing to weight gain. The ethical implications of using these drugs for cosmetic purposes, independent of medical need, further complicate the discussion. As research continues and access to these medications expands, careful consideration of both the individual and societal implications will be crucial.